Our Partners

Contact
67000 Strasbourg
FRANCE
contact@odimma-therapeutics.com
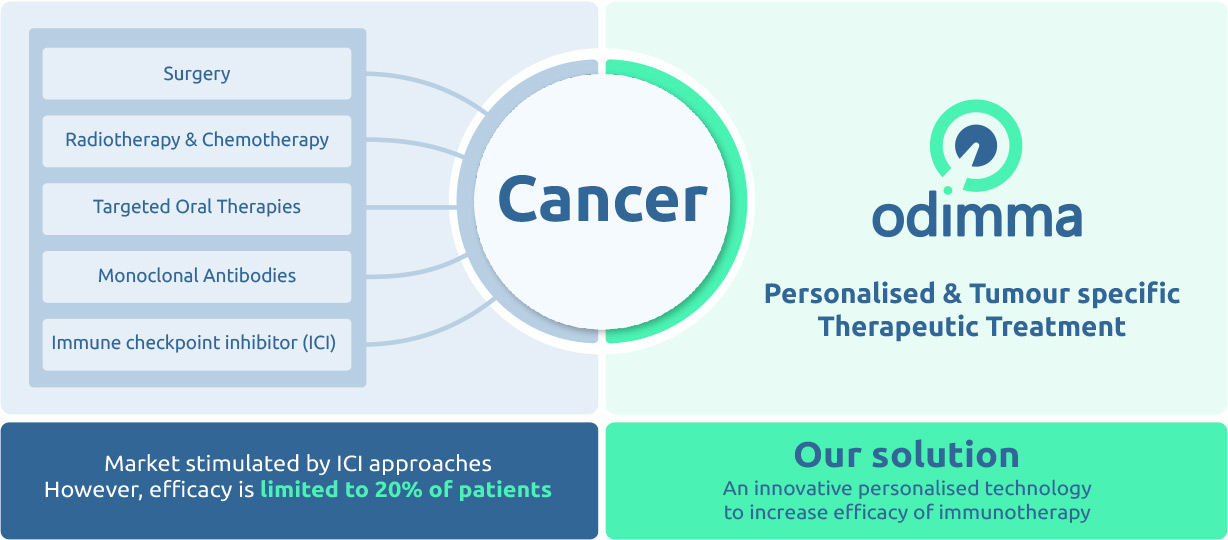
One of the major hurdles in the fight against cancer is that each tumour is genetically unique
and has specific tumour markers.
Odimma has designed a next-generation precision immunotherapy platform
able to induce a powerful cellular immune response against specific tumours markers
as well as a strong anti-tumoural effect in several aggressive models such as melanoma, colorectal and breast cancer.
By harnessing the ability of the patient’s own immune system
to specifically recognise specific markers displayed by tumour cells,
Odimma’s approach represents a game changer in the field of immune oncology
and opens new avenues to fight the most hard-to-treat cancers.

